Ozempic Faces Thousands of Lawsuits Over Severe Stomach Paralysis and Vision Loss, FDA Issues Warning
Thousands of people across the United States have filed lawsuits against Novo Nordisk, the maker of Ozempic, claiming the widely used drug has caused severe and sometimes life-altering side effects.
According to federal court records, more than 3,500 cases are now part of a growing multidistrict litigation (MDL), with approximately 3,546 lawsuits pending as of early April 2026. Legal experts say that number is expected to increase as more patients come forward.
Table of Contents
Why This Matters
Ozempic, a prescription medication originally approved to treat type 2 diabetes, has quickly gained popularity for weight loss. Millions of Americans now use the drug or similar medications.
While many patients report positive results, the surge in lawsuits is raising concerns about the drug’s long-term safety, especially as its use expands beyond diabetes treatment.
Key Allegations
The lawsuits focus on semaglutide, the active ingredient in Ozempic, and other similar drugs known as GLP-1 receptor agonists. Plaintiffs argue that the manufacturer did not provide adequate warnings about serious risks.
Reported Side Effects Include:
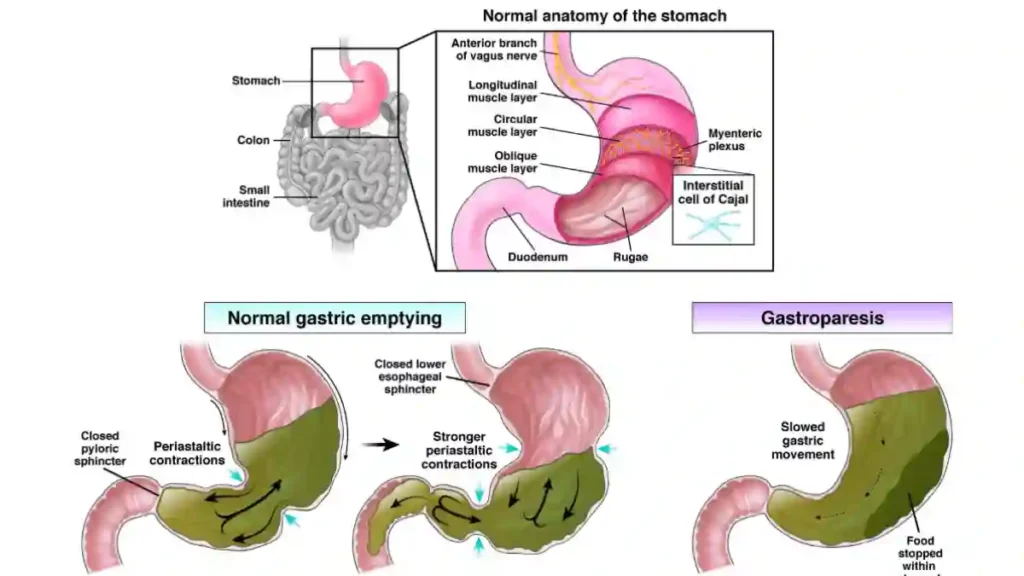
- Severe digestive problems:
Patients claim they developed conditions such as gastroparesis (stomach paralysis), intestinal blockages, chronic nausea, and persistent vomiting. In some cases, these issues required hospitalization and long-term care.
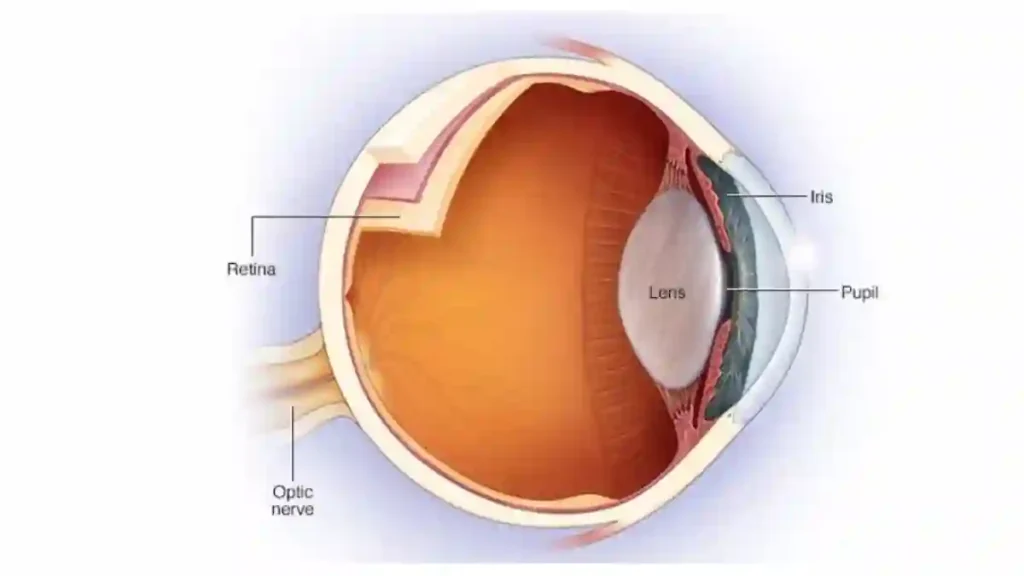
- Vision loss:
Some lawsuits allege sudden vision impairment caused by a condition known as non-arteritic anterior ischemic optic neuropathy (NAION). This condition can result in permanent vision loss in one eye.
Legal proceedings have been divided into separate tracks. The main MDL, which focuses on digestive complications, is being handled in the Eastern District of Pennsylvania. Meanwhile, vision-related claims are being reviewed independently.
Recent court rulings now require some plaintiffs to provide medical evidence, such as gastric emptying tests to support their claims.
FDA Warning and Regulatory Oversight
In March 2026, the U.S. Food and Drug Administration issued a warning letter to Novo Nordisk. The agency stated that the company failed to properly report certain serious side effects, including some cases involving deaths linked to semaglutide.
The FDA also cited delays in reporting other complications, such as strokes and suicidal thoughts.
This is not the first time regulators have raised concerns. In 2023, the FDA required an updated warning label to include the risk of intestinal blockage (ileus).
Despite these issues, regulators say the drug can still be beneficial for approved uses, noting that its advantages may outweigh risks for many patients when used properly. However, officials continue to monitor its safety closely.

Mixed Findings from Recent Research
Not all studies paint a negative picture of the drug.
A large study conducted in Sweden and published in The Lancet Psychiatry in 2026 found that “semaglutide” tablet may actually reduce the risk of worsening mental health conditions. Researchers reported a 42% lower risk of issues such as depression, anxiety, and substance use disorders among certain patients.
Even so, experts caution that stopping the medication can reduce its benefits, particularly for weight loss and heart health. Patients are strongly advised to consult their doctors before making any changes.
Background on Ozempic
Ozempic was approved by the FDA in 2017 for managing type 2 diabetes. It works by mimicking a natural hormone that helps regulate blood sugar and appetite.
In recent years, it has become widely used for weight loss, even outside its original approval. Other drugs in the same category include Wegovy and Mounjaro, developed by Eli Lilly.
Common side effects include nausea and vomiting, but the lawsuits focus on rarer and more severe complications.
Patient Experiences and Legal Challenges
Some patients involved in the lawsuits describe dramatic changes to their daily lives. In court filings, individuals have reported being unable to eat normally and experiencing constant nausea or vomiting after starting the medication.
Medical experts say proving a direct link between the drug and these conditions can be difficult. Disorders like gastroparesis can have multiple causes, especially in people with diabetes.
Novo Nordisk has defended its product, stating that Ozempic’s labeling includes appropriate warnings and that both clinical trials and real-world data support its safety when used as directed.

What Comes Next
Key trial dates, known as bellwether trials, are expected later in 2026. These cases will likely influence how future lawsuits are handled and whether settlements may occur.
At this time, no global settlement has been announced.
Patients who experience severe side effects are encouraged to seek medical attention immediately. Experts also recommend discussing any concerns with healthcare providers before starting or stopping the medication.
Medical Disclaimer: This article is for informational purposes only and is not legal or medical advice. Sources include court documents, FDA announcements, and peer-reviewed studies as of April 2026.